Menstrual Pad–Based HPV Testing Opens a Great Way to Screen Cervical Cancer
Cervical cancer remains a leading cause of cancer death among women in many countries despite the proven effectiveness of HPV-based screening. Expanding access through non-invasive, acceptable, and scalable methods is considered essential to achieving global elimination targets.
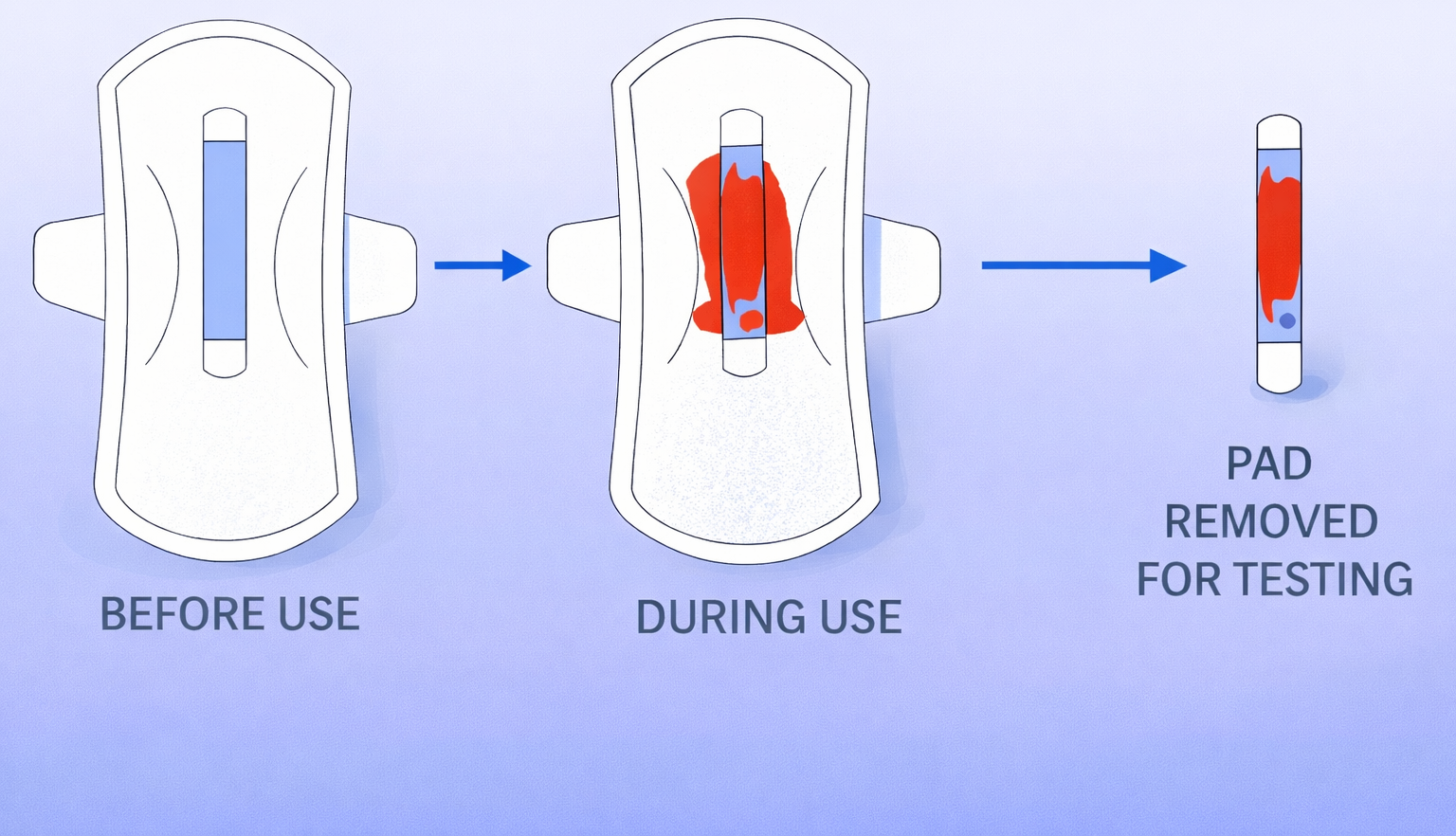
A large population-based study from Hubei Province, China, reports that human papillomavirus (HPV) testing using menstrual blood collected via a specially designed sanitary Mini pad performs comparably to clinician-collected cervical samples for detecting high-grade cervical lesions. The findings support the potential integration of non-invasive menstrual blood testing into routine cervical cancer screening programs.
This cross-sectional study enrolled 3,068 women aged 20–54 years with regular menstrual cycles between September 2021 and January 2025 across four urban and three rural communities. Each participant provided three specimens: menstrual blood collected using a DNA-preserving Mini pad (index test), a clinician-collected cervical sample for HPV testing (comparator test), and a cervical sample for ThinPrep cytology.
Among participants, Mini pad-based HPV testing demonstrated a sensitivity of 94.7% (95% CI 80.9–99.1%) for detecting cervical intraepithelial neoplasia grade 2 or worse (CIN2+), comparable to clinician-collected HPV testing at 92.1% (77.5–97.9%). Specificity was slightly lower for the Mini pad method (89.1% vs. 90.0%; P=0.001), but negative predictive values were identical at 99.9%. Positive predictive values (9.9% vs. 10.4%) and referral efficiency (10.1 vs. 9.6 referrals per CIN2+ detected) were similar between methods.
Menstrual blood–based HPV testing differs from other self-sampling approaches by aligning with women’s natural menstrual cycles and avoiding vaginal insertion. Prior observational studies have reported high concordance between menstrual blood HPV testing and clinician-collected samples, as well as strong participant preference for pad-based collection over vaginal swabs.
While this approach enhances privacy, comfort, and accessibility, particularly for women facing sociocultural stigma, geographic barriers, there are some limitations a) multiple genital sites, broader HPV detection may increase false positives, underscoring the need for improved triage markers and b) diagnostic accuracy estimates were constrained by sample size, particularly for higher-grade lesions, requiring further validation in diverse and higher-risk populations.
Nonetheless, the Hubei study suggests that standardized Mini pad collection, combined with digital platforms for specimen tracking and follow-up coordination, could enhance real-world implementation. Women with dual negative results were scheduled for annual community follow-up, reflecting risk-based management principles. Hence, the menstrual blood-based HPV testing could serve as an equivalent alternative to clinician-based sampling, with non-inferior sensitivity and operational feasibility at the population level.
This technique may represent a practical innovation capable of increasing screening coverage, particularly in communities where conventional pelvic examinations remain a barrier.
Ref:
- Lancet Oncol. 24, e204–e216 (2023). doi: 10.1136/bmj.k4823.
- BMJ Glob. Health 7, e008815 (2022). doi: 10.1136/bmjgh-2018-001351.
- WHO. Global strategy to accelerate the elimination of cervical cancer (2020; progress updates 2023).
- BMJ 2026;392:e084831. doi: 10.1136/bmj-2025-084831.
- Clinical microbiology and infection. 2020 May 1;26(5):579-83.
- Bulletin of the World Health Organization. 2013;91:683-90. https://doi.org/10.2471/BLT.12.116020
- Cancer Epidemiology, Biomarkers & Prevention. 2015 May 1;24(5):769-72. https://doi.org/10.1158/1055-9965.EPI-14-1417
- PloS one. 2017 Sep 5;12(9):e0183924. https://doi.org/10.1371/journal.pone.0183924
- BMC women’s health. 2022 Dec 2;22(1):486. https://doi.org/10.1186/s12905-022-02043-y